When you hear biosimilars, highly similar versions of complex biologic drugs that are not exact copies but proven to work the same way in the body. Also known as biologic generics, they’re not like the simple generic pills you pick up at the pharmacy. These are made from living cells, not chemicals, and they treat serious conditions like cancer, rheumatoid arthritis, and diabetes. Unlike regular generics, which are exact copies of small-molecule drugs, biosimilars are built to match the structure and function of their reference biologics—drugs like Humira, Enbrel, or Remicade—that can cost tens of thousands a year.
The biologics, medications made from living organisms, often proteins or antibodies, used to treat chronic and life-threatening diseases they copy are too complex to replicate perfectly. That’s why regulators like the FDA don’t call them generics—they call them biosimilars, medications that are highly similar to an approved biologic with no clinically meaningful differences in safety or effectiveness. To get approval, manufacturers must prove their version works just as well in clinical trials, with no extra side effects. It’s not about cutting corners—it’s about matching the original with precision.
Why does this matter? Because drug affordability, the ability of patients to access and pay for necessary medications without financial hardship is broken for many. Biologics are expensive not because they’re better, but because patents lock out competition for years. Biosimilars break that lock. In the U.S., they’ve already saved billions. In Europe, where they’ve been around longer, they’ve cut prices by 30% to 80%. That means more people get treatment, not just those with top-tier insurance.
But it’s not all smooth sailing. Some doctors still hesitate to switch patients from the original biologic, even when guidelines say it’s safe. Others worry about subtle differences in how the body reacts—especially for drugs with a narrow therapeutic window. And while biosimilars are cheaper, they’re still pricey compared to pills. Still, the trend is clear: more biosimilars are coming. The FDA has approved dozens, and hundreds more are in the pipeline. If you’re on a biologic right now, you might soon have a lower-cost option.
What you’ll find below are real stories and facts about how biosimilars fit into the bigger picture of medication safety, access, and cost. You’ll read about how patent battles delay their arrival, how some patients react differently to them, and why even small changes in inactive ingredients can matter. You’ll also see how they connect to broader issues like generic drug approval, drug shortages, and patient assistance programs. This isn’t theoretical—it’s happening in clinics, pharmacies, and living rooms right now. Whether you’re a patient, caregiver, or just trying to understand your bill, these posts give you the facts you need to ask the right questions.

Understand how 2025 Medicare Part D formulary updates are forcing generic and biosimilar switches, what drugs are affected, and how to protect your coverage before January 1.
Biosimilars are the closest thing to generics for complex biologic drugs. They're highly similar, FDA-approved, and can save patients up to 60% on costs. Learn how they work, why they're not exact copies, and how to use them safely.
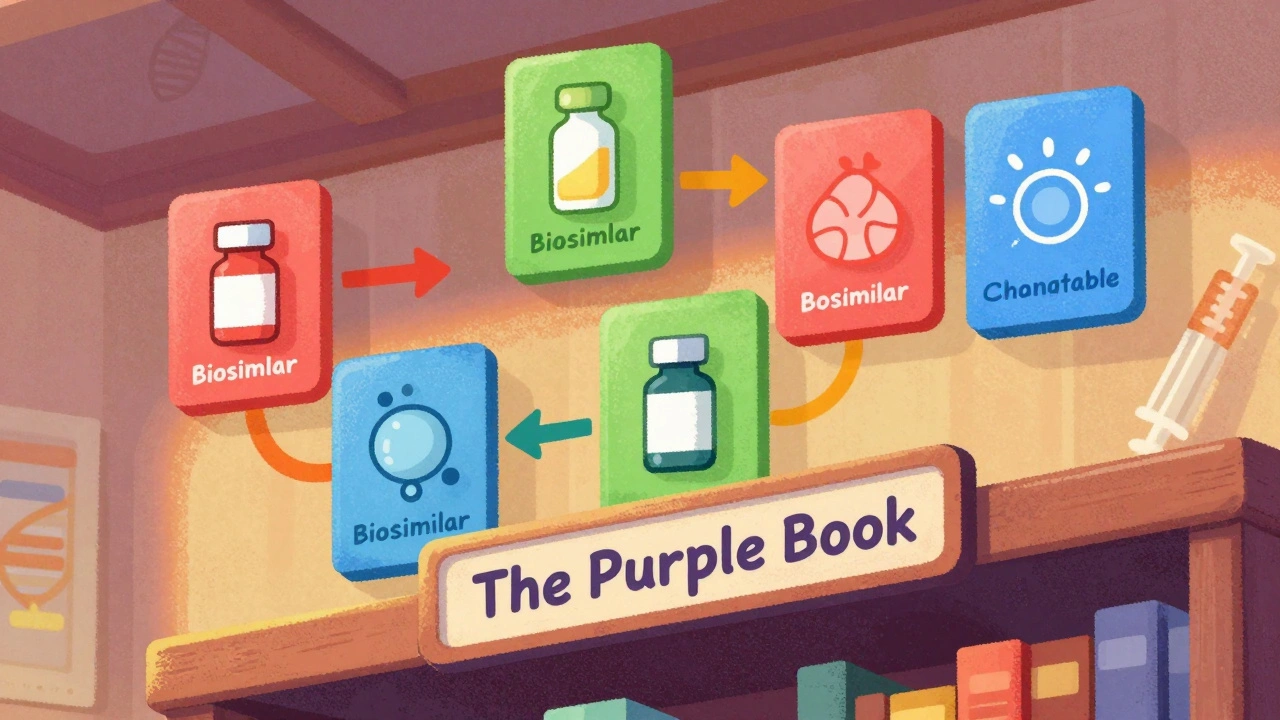
The FDA's Purple Book is the official guide to biosimilars and interchangeable biological drugs. Learn how it works, what the difference is between biosimilars and interchangeable products, and how pharmacists use it to make safe substitutions.
Coronary artery disease, caused by atherosclerosis, is the leading cause of death worldwide. Learn how plaque builds up, who’s at risk, and what treatments actually work-based on the latest 2023 guidelines.

Explore how common performance‑enhancing drugs affect athletes' health, the key side effects, real‑world data, and safe alternatives for optimal performance.

Military deployment exposes medications to extreme heat, storage failures, and access delays that can render life-saving drugs ineffective. From vaccines to insulin, improper storage threatens soldier readiness-and the military is racing to fix it.

Fluoxetine, an antidepressant, is often prescribed to treat depression, anxiety, and various mood disorders. Understanding its medical effects, potential side effects, and interactions with other medications can help users make informed decisions. This article also covers common dosages and recommendations, ensuring safety and effectiveness. Discover ways to access the best deals with practical tips and considerations.

Prescription assistance programs from drug manufacturers help millions afford costly medications. Learn how copay cards and PAPs work, who qualifies, what you can save, and the hidden rules that could block your help.