When navigating clinical trials, systematic studies that test medical interventions in humans. Also known as human studies, they create the evidence doctors rely on every day. clinical trials aren’t just paperwork; they are the bridge between a new molecule in the lab and a treatment you can actually use. A trial always starts with a clear objective, whether it’s proving a drug works better than the current standard or confirming that a novel therapy is safe enough to move forward. This goal sets up the entire design, from who can join to how the data will be measured.
Understanding drug efficacy, the degree to which a medication achieves its intended therapeutic effect in patients is the heart of any trial. Researchers compare the new drug’s performance against a control—often a placebo or an existing treatment—to see if the benefits outweigh any risks. That comparison creates a semantic triple: clinical trials evaluate drug efficacy. When the study is a randomized controlled trial, a design where participants are randomly assigned to treatment or control groups, the results gain statistical power because randomization reduces bias. This structure yields another triple: randomized controlled trials require random assignment. The placebo, defined as placebo, an inactive substance used to blind participants and researchers, plays a crucial role in blinding, ensuring participants don’t know which arm they’re in, which in turn protects the integrity of efficacy data.
Every trial also hinges on patient enrollment, the process of recruiting and screening eligible volunteers for a study. Without enough qualified participants, even the best‑designed study can’t reach statistical significance. Enrollment ties directly to trial feasibility, influencing timelines and budget. Moreover, diverse enrollment improves the generalizability of findings across age groups, ethnicities, and comorbid conditions. This creates the third triple: clinical trials depend on patient enrollment. Researchers must balance strict inclusion criteria—aimed at safety and clear results—with the need for a sample that reflects real‑world patients.
The articles below dive deep into specific trials, drug comparisons, and safety reviews that illustrate these concepts in action. From how mefenamic acid impacts bone health to the latest insights on hepatitis C fatigue, each piece highlights a slice of the trial ecosystem—showcasing design choices, outcomes, and practical takeaways for clinicians and patients alike. As you explore, you’ll see how the principles of efficacy, randomization, placebos, and enrollment shape the evidence that guides everyday medical decisions.

Explore the latest research, new formulations, and future directions for metoclopramide, including safety updates, combination therapies, and personalized dosing.
Explore Avanafil's journey from discovery to FDA approval, covering research, trials, regulatory hurdles, and its impact on erectile dysfunction treatment.
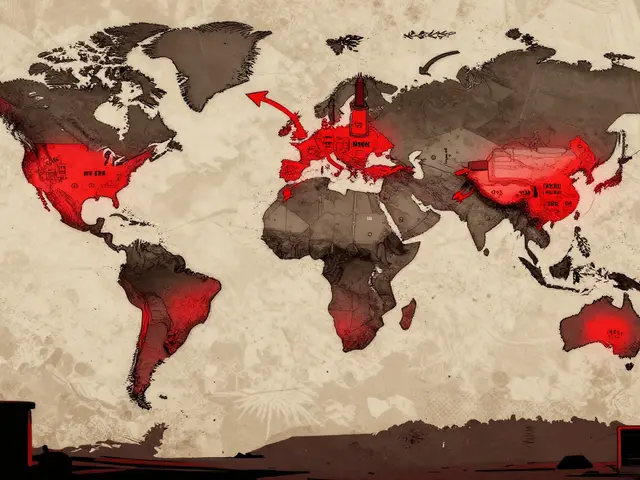
Dependence on foreign manufacturing for pharmaceutical ingredients is causing widespread drug shortages. With over 80% of active ingredients coming from just a few countries, disruptions in China or India directly impact patient access to life-saving medications.

Learn what Rhinocort does, how to spray it correctly, dosage guidelines, common side effects and alternatives for allergy relief.

Learn how drug take-back programs safely dispose of unused medications through permanent drop boxes, mail-back envelopes, and nationwide events. Find out what you can and can’t dispose of, and how to locate the nearest collection site.

Warfarin and DOACs are both blood thinners used to prevent clots, but DOACs are now preferred for most patients due to better safety, fewer interactions, and no need for regular blood tests. Learn the key differences and what to do in an emergency.

Learn how to buy Naprosyn online in the UK, discover safe pharmacies, current regulations, and key tips for purchasing Naproxen online in 2025.