
Most people don't tell their doctors about dietary supplements-even though they can interact dangerously with medications. Learn why full disclosure matters, which supplements are riskiest, and how to talk to your care team safely.
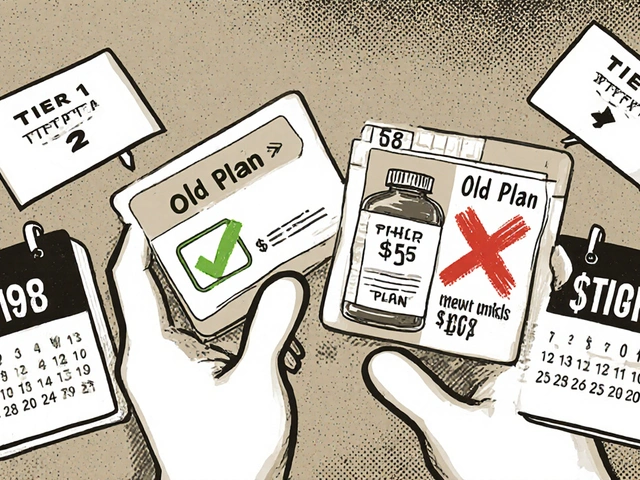
When switching health plans, your generic drug coverage can save or cost you hundreds a year. Learn how formulary tiers, deductibles, and state rules impact your prescription costs-and how to avoid expensive surprises.

Fiber supplements like psyllium help with constipation but can interfere with medications. Learn the safest timing rules to avoid reduced drug effectiveness and side effects.

Learn practical diet and nutrition strategies to keep estradiol levels balanced, covering phytoestrogens, omega‑3s, vitamin D, fiber and more.
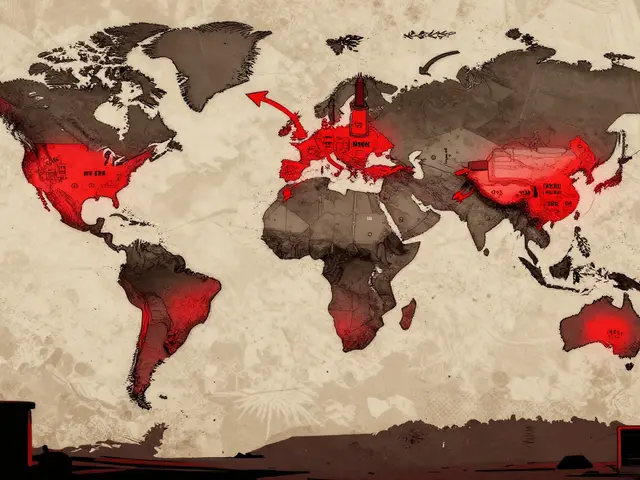
Dependence on foreign manufacturing for pharmaceutical ingredients is causing widespread drug shortages. With over 80% of active ingredients coming from just a few countries, disruptions in China or India directly impact patient access to life-saving medications.
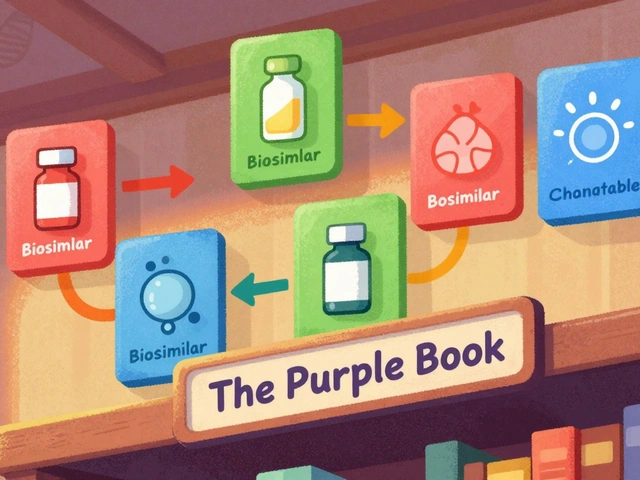
The FDA's Purple Book is the official guide to biosimilars and interchangeable biological drugs. Learn how it works, what the difference is between biosimilars and interchangeable products, and how pharmacists use it to make safe substitutions.