When a non-compliant drug manufacturer, a company that fails to meet federal standards for drug production, safety, or labeling. Also known as unregulated pharmaceutical producer, it can release medicines that are ineffective, contaminated, or even dangerous. These aren’t just minor mistakes—they’re serious violations that can lead to hospitalizations, overdoses, or deaths. The FDA doesn’t just inspect factories once a year. They send teams unannounced, check records down to the batch number, and track every ingredient from raw material to finished pill. But some companies still cut corners—because it’s cheaper, faster, and sometimes, they think they won’t get caught.
What makes a manufacturer non-compliant? It’s not just about dirty floors. It’s about FDA regulations, the strict rules that govern how drugs are made, tested, and labeled in the U.S.. Think of it like baking a cake: if you swap out the flour for sawdust, it might look the same—but it won’t work. Some manufacturers substitute active ingredients with cheaper fillers, skip stability testing, or fake lab results. Others import raw materials from unapproved suppliers, or don’t clean equipment between batches, risking cross-contamination. These are the same issues that show up in recalls for generic drug safety, the risk that cheaper versions of brand-name drugs may not meet the same quality standards. And it’s not rare. The FDA has issued dozens of warning letters to foreign and domestic plants in the last five years alone.
Why does this matter to you? Because if your blood pressure pill doesn’t dissolve properly, or your antibiotic is 30% weaker than it should be, your health is on the line. You might not notice right away—but over time, it adds up. A study from 2022 found that nearly 1 in 5 generic drug recalls were tied to manufacturing violations, not just packaging errors. And when a company ignores pharmaceutical quality control, the systematic process of ensuring drugs are consistently produced and controlled according to quality standards, patients pay the price. That’s why it’s not just about trusting your pharmacy—it’s about knowing that the system is working.
What you’ll find in these posts isn’t just a list of bad companies. It’s a look at how these failures connect to real-world problems: why some people react badly to generics, how patent tricks delay cheaper drugs, why compounding pharmacies sometimes fill gaps when safe ones aren’t available, and how the FDA’s own rules can be manipulated. These aren’t abstract policy debates—they’re about whether your next pill will actually work.

The FDA uses Import Alerts to automatically block drug shipments from non-compliant manufacturers. With the 2025 Green List initiative, enforcement against GLP-1 APIs has become the strictest in history-reshaping global supply chains and raising compliance standards.

FDALabel is the FDA's free, searchable database of over 149,000 drug labels. Learn how to search by section, pharmacologic class, and MedDRA terms to find accurate drug safety information for research, compliance, and clinical use.

Explore how medication helps manage fibromyalgia pain, the main drug classes, their benefits, side effects, and tips for choosing the right regimen.
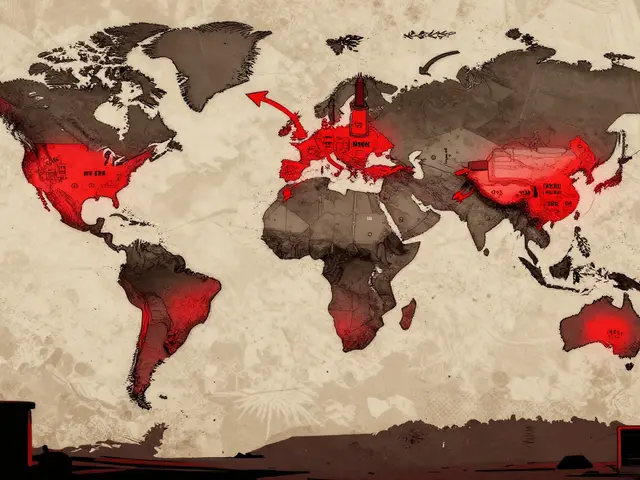
Dependence on foreign manufacturing for pharmaceutical ingredients is causing widespread drug shortages. With over 80% of active ingredients coming from just a few countries, disruptions in China or India directly impact patient access to life-saving medications.

A detailed side‑by‑side comparison of Neoral (Cyclosporine) with Tacrolimus, Sirolimus, Mycophenolate, and Azathioprine, covering mechanisms, dosing, side effects, cost and monitoring.
Learn how to differentiate between generic and authorized generic medications to reduce prescription costs. This guide explains insurance formularies, PBM rebates, and specific questions to ask your pharmacist.