When you hear interchangeable biosimilars, a type of biologic medication approved by the FDA to be substituted for the original brand drug without needing a doctor’s reauthorization. Also known as biosimilar substitutes, they’re not just cheaper copies—they’re scientifically matched versions of complex, living-cell-derived drugs like Humira or Enbrel. Unlike regular generics, which are simple chemical copies, biosimilars come from living organisms, making them far harder to replicate exactly. But when a biosimilar gets the "interchangeable" label, it means the FDA has confirmed you can switch between it and the original drug as easily as you would between two brands of aspirin—no extra paperwork, no doctor’s note, no risk.
This matters because biologics can cost over $100,000 a year. Many patients can’t afford them, and insurers won’t cover them without alternatives. biosimilars, medications that are highly similar to an already-approved biologic but not identical. Also known as biologic generics, they’ve been around since 2015, but only a handful have reached the interchangeable status. That’s changing fast. The FDA has been tightening its standards, requiring real-world data showing no increased risk of side effects or reduced effectiveness when patients switch. FDA biosimilar approval, the process by which the U.S. Food and Drug Administration evaluates and authorizes biosimilar and interchangeable biologics for market. Also known as biologics license application pathway, it’s now the key gatekeeper for cost savings across diabetes, rheumatoid arthritis, and cancer treatments. And while some doctors still hesitate to switch patients, studies show no difference in outcomes when patients are moved from Humira to its interchangeable biosimilar—over 10,000 patients tracked across multiple trials.
But here’s the catch: not all biosimilars are interchangeable. Many are approved as "biosimilar" only, meaning your pharmacist can’t swap them without your doctor’s okay. That’s why understanding the difference matters when you’re filling a prescription. It’s also why some patients report side effects after switching—sometimes it’s the inactive ingredients, sometimes it’s the nocebo effect, and sometimes it’s just bad luck with a particular batch. The drug substitution, the practice of replacing a prescribed medication with a chemically or biologically similar alternative. Also known as pharmaceutical substitution, it’s the backbone of savings in modern healthcare. But it only works if you trust the system. And that’s why the posts below dig into real cases: why some people react differently to biosimilars, how the FDA tracks safety after approval, what happens when a drug is switched mid-treatment, and why some insurers still block the cheapest option—even when it’s officially interchangeable.
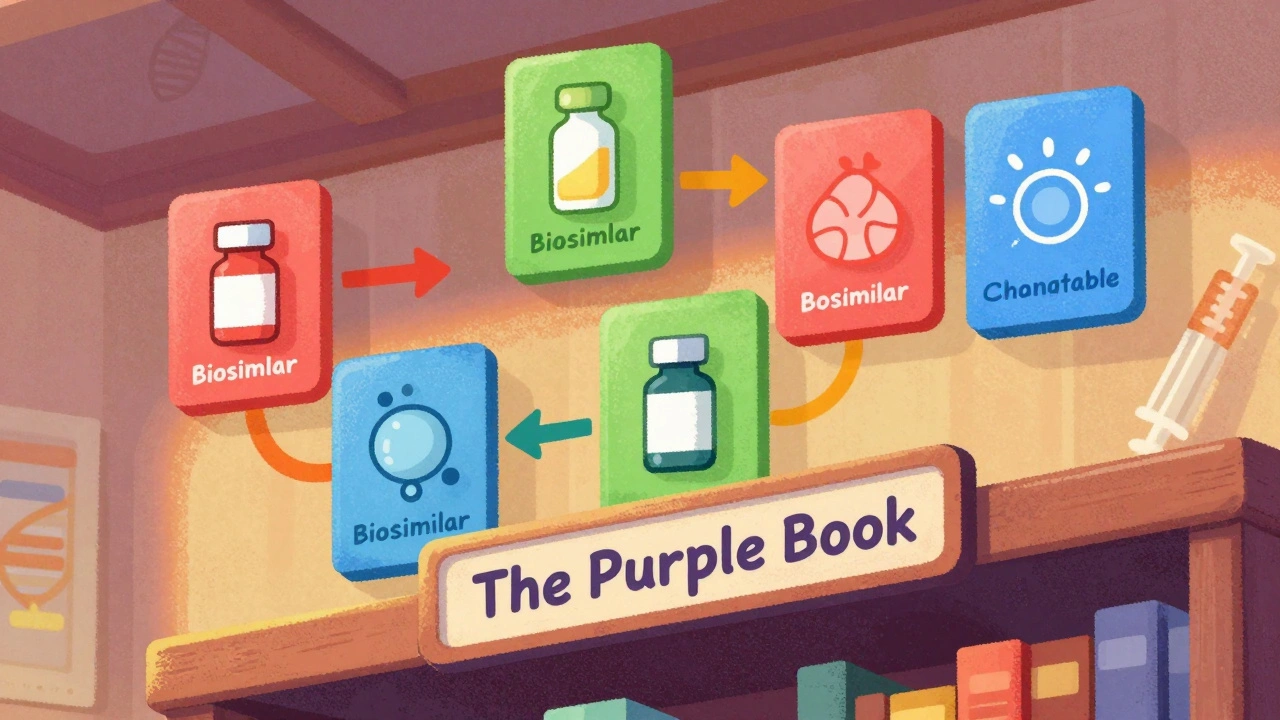
The FDA's Purple Book is the official guide to biosimilars and interchangeable biological drugs. Learn how it works, what the difference is between biosimilars and interchangeable products, and how pharmacists use it to make safe substitutions.

Learn how to manage gestational diabetes with diet, exercise, and blood sugar monitoring to reduce risks during pregnancy and protect long-term health for both mother and baby.

Most people don't tell their doctors about dietary supplements-even though they can interact dangerously with medications. Learn why full disclosure matters, which supplements are riskiest, and how to talk to your care team safely.
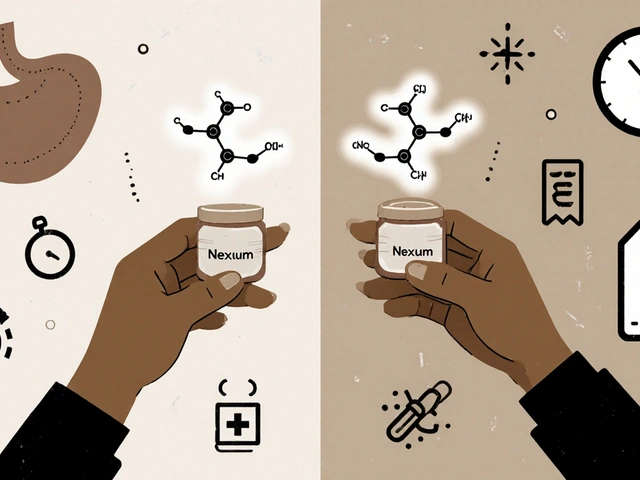
Compare Nexium (esomeprazole) with generic alternatives like omeprazole, lansoprazole, and pantoprazole. Learn which PPI works best for acid reflux, how to switch safely, and when lifestyle changes can replace medication.

Prescription discount programs like GoodRx and manufacturer coupons can slash medication costs - but only if you use them right. Learn who saves the most, when they backfire, and how to avoid costly mistakes.
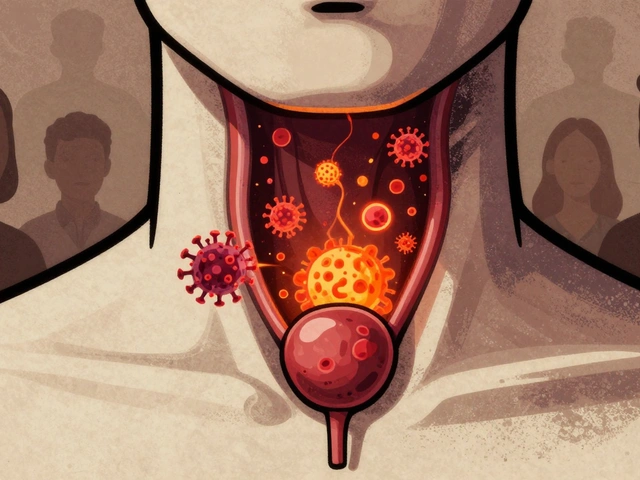
HPV causes thousands of throat and anal cancers each year in the U.S., with rising rates among men. Vaccination is the most effective way to prevent these cancers - yet coverage remains too low. Learn how HPV leads to cancer and what you can do to stop it.