When a brand-name drug’s patent runs out, the 180-day exclusivity, a legal incentive granted by the FDA to the first generic manufacturer to challenge a patent and win. Also known as generic drug market exclusivity, it’s meant to speed up competition and lower prices—but it doesn’t always work that way. This rule lets one generic company be the only seller for six months after approval, giving them a chance to capture market share before others jump in. Sounds fair, right? But here’s the catch: sometimes that first company doesn’t lower prices at all. They might even raise them, knowing they have a monopoly for half a year.
This system ties directly to FDA approval, the process that validates generic drugs as safe and effective replacements for brand-name versions. Also known as ANDA pathway, it’s how generics get legal clearance without repeating expensive clinical trials. But the pharmaceutical patents, legal protections that give drugmakers exclusive rights to sell a medication for a set number of years. Also known as brand-name drug monopolies, they’re the reason 180-day exclusivity even exists. When a patent expires, the race begins: who files first? Who challenges the patent successfully? Who gets the exclusive window? The answer shapes whether you pay $10 or $100 for the same pill.
It’s not just about who gets in first—it’s about who gets left out. Some companies file for exclusivity just to block others, then sit on it. That’s called evergreening—a legal loophole where the first generic doesn’t even launch, and no one else can enter. You’ve probably seen this with drugs like Lipitor or Nexium: brand names stay expensive long after patents should’ve expired. Meanwhile, patients wait, doctors scramble, and pharmacies can’t stock cheaper versions.
The posts below show how this system plays out in real life. You’ll find stories about why some generic versions of levothyroxine or warfarin still cause problems for patients, even when they’re FDA-approved. You’ll see how government policies and market forces try to keep prices down—without direct price controls. And you’ll learn how prescription assistance programs and health plan formularies try to fill the gaps when exclusivity fails.
180-day exclusivity isn’t a villain. It was designed to help. But like any rule without oversight, it can be twisted. What matters is what happens after the patent expires—whether competition actually kicks in, or whether your wallet pays the price instead.

The 180-day exclusivity rule under the Hatch-Waxman Act was meant to speed up generic drug entry-but now it often delays it. Learn how patent challenges, FDA rules, and corporate strategy keep prices high and patients waiting.

Fiber supplements like psyllium help with constipation but can interfere with medications. Learn the safest timing rules to avoid reduced drug effectiveness and side effects.
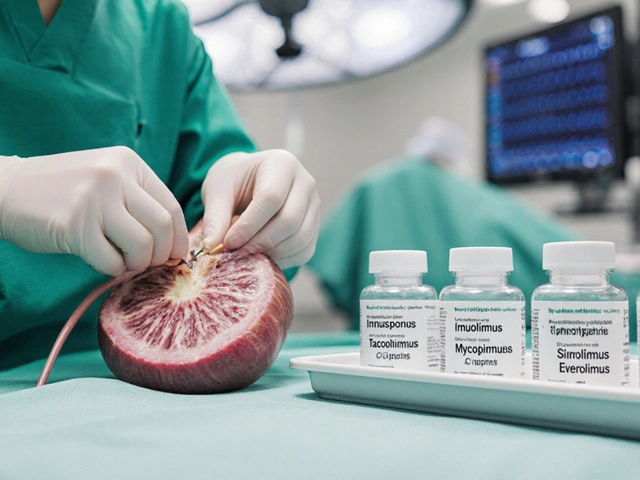
A side‑by‑side look at Imusporin (cyclosporine) versus tacrolimus, mycophenolate, sirolimus, everolimus and others, covering mechanisms, side effects, UK costs and when to switch.
Explore the latest research, new formulations, and future directions for metoclopramide, including safety updates, combination therapies, and personalized dosing.

Learn what Rhinocort does, how to spray it correctly, dosage guidelines, common side effects and alternatives for allergy relief.

Learn step‑by‑step how to find reputable online pharmacies, understand price factors, and use generic amoxicillin safely while avoiding counterfeit risks.