When you pick up a generic pill at the pharmacy and pay a fraction of what the brand name costs, you’re seeing the result of an ANDA, Abbreviated New Drug Application, the FDA’s official pathway for approving generic versions of brand-name medications. Also known as Abbreviated New Drug Application, it’s the quiet engine behind most of the affordable medicines in your medicine cabinet. Unlike brand drugs that need full clinical trials to prove safety and effectiveness, generics use the existing data from the original drug—saving time, money, and ultimately, your wallet.
That’s not magic. It’s regulation. The FDA, U.S. Food and Drug Administration, the federal agency responsible for ensuring the safety and efficacy of drugs and medical products doesn’t just approve generics blindly. Every ANDA must prove the generic version is bioequivalent—meaning it works the same way in your body, at the same dose, with the same results. No guesswork. No shortcuts. And because dozens of companies can file an ANDA for the same drug, competition kicks in fast. That’s why a 30-day supply of generic lisinopril might cost $4 instead of $150. It’s not a sale. It’s how the system was built to work.
Behind every cheap generic is a chain of oversight: the FTC, Federal Trade Commission, the U.S. agency that enforces antitrust laws and prevents anti-competitive practices in the marketplace watching for collusion, manufacturers being audited for quality, and pharmacies switching between suppliers based on price. This isn’t theoretical. It’s why you might get a different-looking pill from the same generic brand next month—and why it still works just as well. The system works because it’s designed to be transparent, competitive, and patient-focused.
You’ll find posts here that dig into how governments use this system to keep prices low without setting caps, how switching health plans affects your access to these approved generics, and even how storage and interactions with these same drugs can make or break your treatment. Whether you’re comparing generic tadalafil options or trying to understand why your ramipril costs less than it did last year, the answer always ties back to ANDA. This isn’t just a form. It’s the reason you can afford to stay healthy.

Generic drugs contain the same active ingredients as brand-name medications and are just as safe and effective. They cost up to 85% less because they don't repeat expensive clinical trials. Learn how they're approved, why they're different in appearance, and when to choose them.

Sunburn and heatstroke often strike during hot days but are not the same thing. This article spotlights key differences, how to recognize early warning signs, and real-life ways to treat each condition if you or someone else gets hit. It covers prevention tips, real facts, and the science behind both sunburn and heatstroke. Whether you're sunbathing, working outdoors, or just enjoying a sunny weekend, you'll find clear guidance to keep yourself safe. Read on to know what to do if the summer heat gets the better of you.
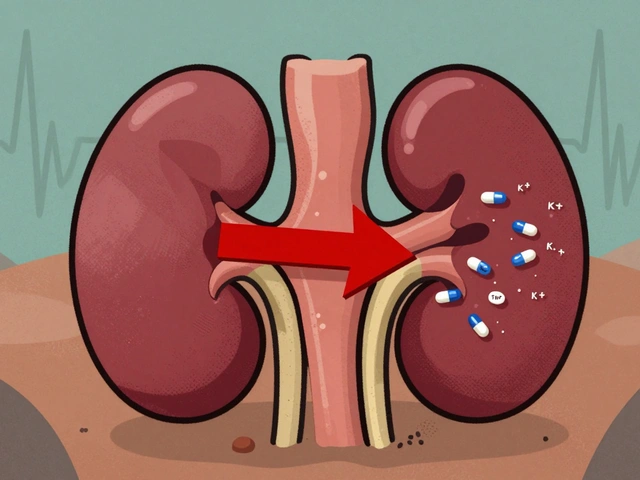
Trimethoprim can cause dangerous spikes in potassium levels, especially in older adults or those on blood pressure meds. Learn who's at risk, how to prevent life-threatening hyperkalemia, and safer antibiotic alternatives.

Learn practical ways to boost iron, B12, calcium, vitamin D and zinc absorption on vegetarian and vegan diets with food combos, cooking hacks, and gut‑health tips.

A practical guide comparing Modvigil (modafinil) with armodafinil, adrafinil, Provigil and caffeine‑L‑theanine, covering cost, duration, safety and best use cases.

Generic drugs save Americans over $445 billion annually, making up 90% of prescriptions but only 12% of drug spending. Learn the real numbers behind generic vs. brand-name drug costs and how to save on prescriptions.